Surface free energy (SFE), surface energy
Surface free energy (SFE) is the work that would be necessary to increase the surface area of a solid phase. SFE has a decisive influence on the wettability of solids by liquids. It is therefore an important parameter for the optimization of coating processes, but also for any other type of solid-liquid contact.
When does one speak of SFE, when of surface tension?
The terms SFE and surface tension (SFT) are physically equivalent. SFE is usually used for solid surfaces and SFT for liquid surfaces. Occasionally, however, the SFT of a solid is also referred to.
The SFE has the unit mJ/m2 (millijoule per square meter) as the energy per area, whereby the equivalent unit mN/m (millinewton per meter), which is commonly used for the SFT, is also frequently used. The formula symbol is σ (small sigma), more rarely γ (small gamma).
The word "free" indicates that this is the part of the energy that can be converted into mechanical work, in contrast to the internal energy, which also contains the heat-related entropy. In practice, the term "free" is often omitted.
What is the connection between SFE and wettability?
Every system strives for a state of free energy that is as low as possible. Liquids therefore take the smallest possible surface area at a given volume due to the SFT; in weightlessness they form spherical droplets. However, solids cannot minimize their surface by deformation, but they can form an interface with a liquid to reduce free energy, i.e. they can be wetted. Therefore, the SFE of a solid is closely related to its wettability.
How can SFE be influenced?
Good wettability and a correspondingly high SFE are required, for example, when bonding, coating, or printing. In other areas, such as corrosion and moisture protection, wettability must be reduced. A large number of technical processes prepare solid surfaces for contact with liquids - most of which directly or indirectly alter the SFE.
Increasing the SFE is of central importance for plastic surfaces. The best known methods are plasma, flame and corona treatment, as well as chemical processes with oxidizing agents. Industrial cleaning removes low-energy contamination by fats or oils. The surface then shows a higher SFE.
A low SFE and correspondingly low wetting is usually achieved by coating with low-energy substances. Examples are PTFE-coated cooking utensils or the use of oils for corrosion protection.
How are the SFE and the contact angle related?
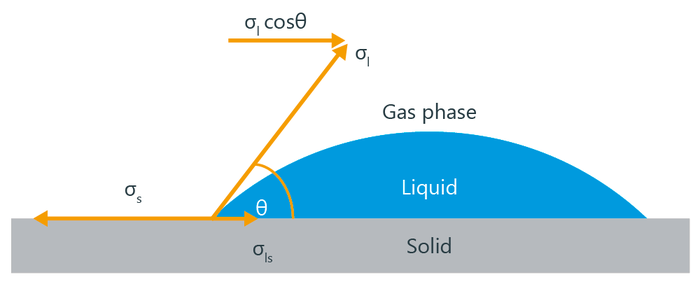
The measure of wettability is the contact angle (CA) θ (small theta), which is usually determined optically as the angle at the intersection of the contour of a drop with the plane of the surface (= baseline). According to Young's equation, the CA results from a force equilibrium of three tension or energy components, each of which strives to minimize the surface or interface:
- The SFE of the solid, σs
- The SFT of the liquid, σl
- The interfacial tension (IFT) between solids and liquids, σls
Young's equation for the relation of these components is:
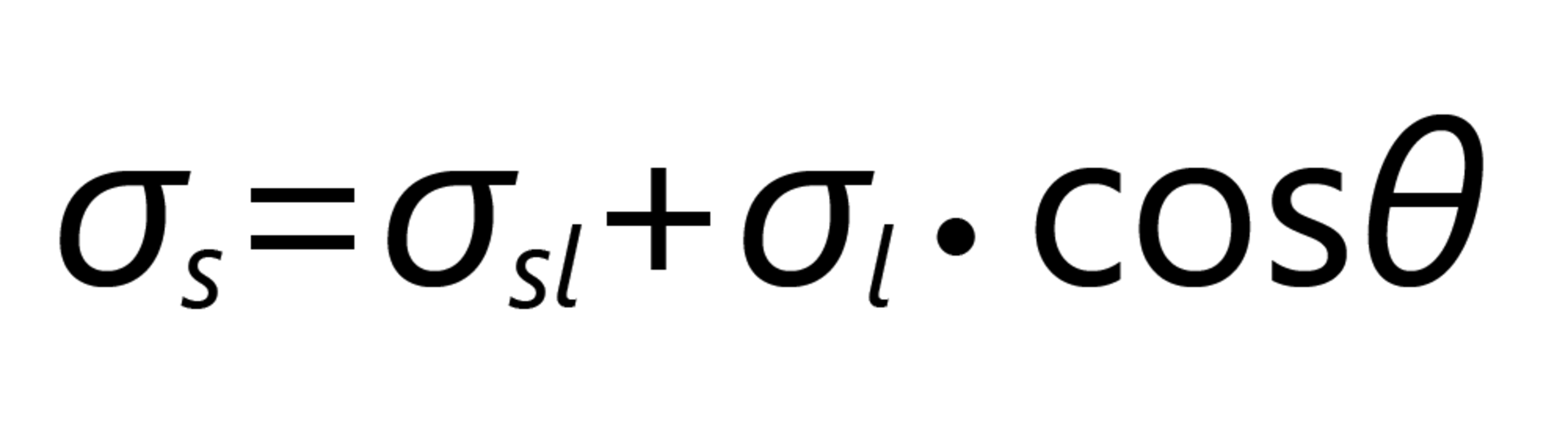
The following illustration shows the contact angle resulting from the equilibrium of forces during solid wetting according to Young:
How is the SFE calculated from contact angle data?
If the CA is measured with a liquid of known SFT, then two quantities of Young's equation remain unknown: the sought SFE and the IFT. The key to the solution therefore lies in the description of the IFT.
If there were no interactions between the liquid and the solid surface in the two-phase contact, they would behave like two separate surfaces. The IFT would then be the sum of SFE and SFT; the contact angle in this theoretical case would be 180° (cos θ =-1).
If interactions occur between the phases, which is always the case in practice, the IFT is reduced by the energy contribution resulting from the interactions:
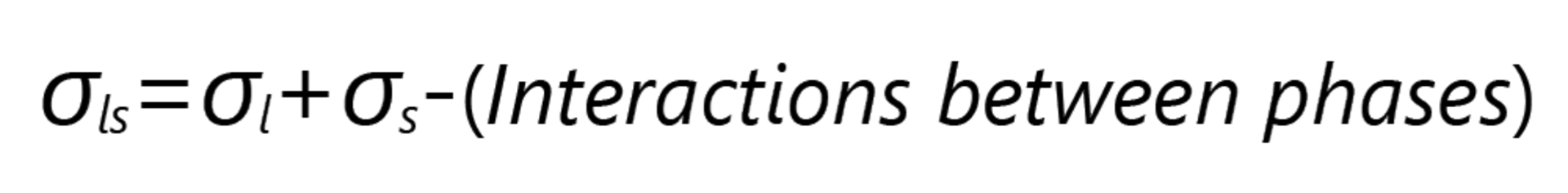
There are different models for calculating SFE, which differ mainly in the interpretation and calculation of these interfacial interactions. The most common is the subdivision between polar and disperse interaction fractions, where the SFE and the SFT are split into a polar and a disperse fraction. Simplifying it is assumed that only similar interactions take place between the phases, so that polar only with polar and disperse only with disperse fractions of the adjacent phases are put into relation with each other.
In order to determine the SFE, contact angles are measured with at least two liquids in which both the SFT and their polar and disperse fractions are known. To calculate the SFE, Young's equation is combined with equations to calculate the interactions.
What are polar and disperse fractions of SFE?
The relatively strong polar interactions are caused by permanent and localizable asymmetry of electron density in molecules. In liquids, the best known example is water, the polarity of which is responsible for the high SFT. Glass is a typical example of a strongly polar solid surface.
Disperse interactions are usually weaker; they result from statistical fluctuations of the electron density distribution in a molecule, which cause temporary charge differences at different locations. This leads to electrostatic attraction between molecules. Alkanes and some plastics such as polyethylene or polypropylene exclusively form disperse interactions. This is also the reason for the poor wettability of many plastics by water.
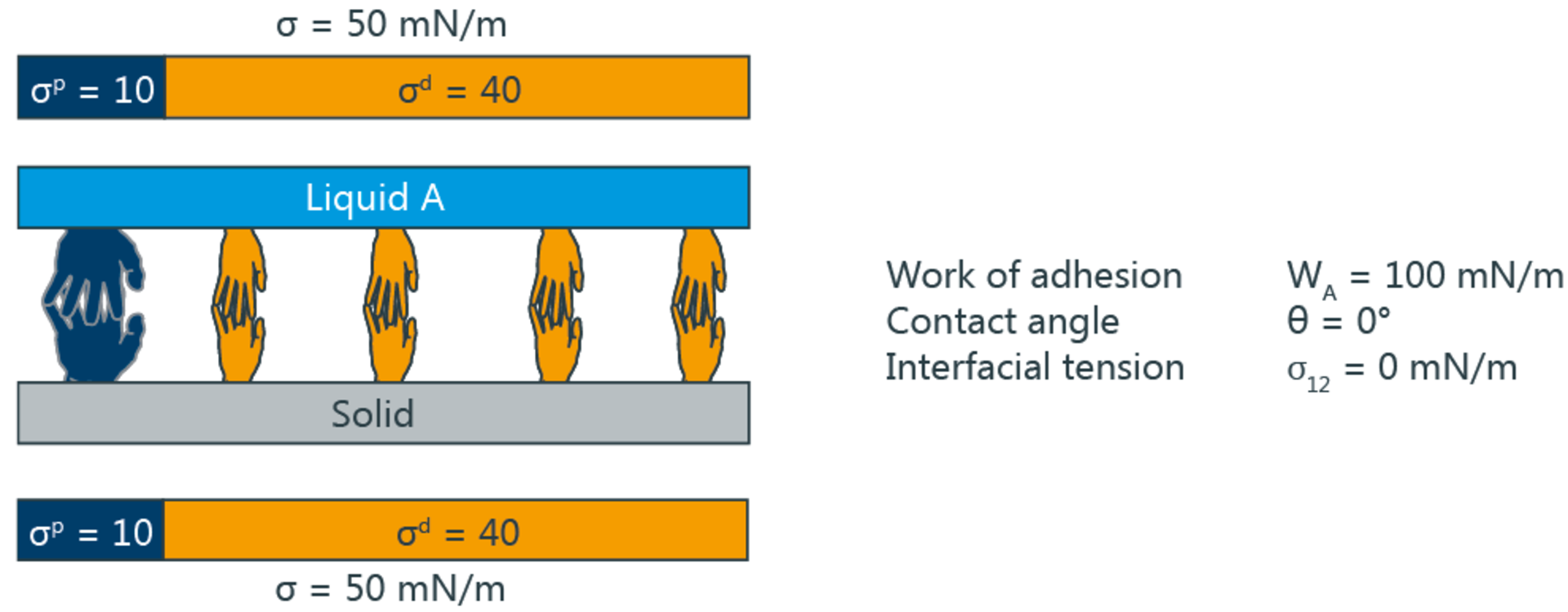
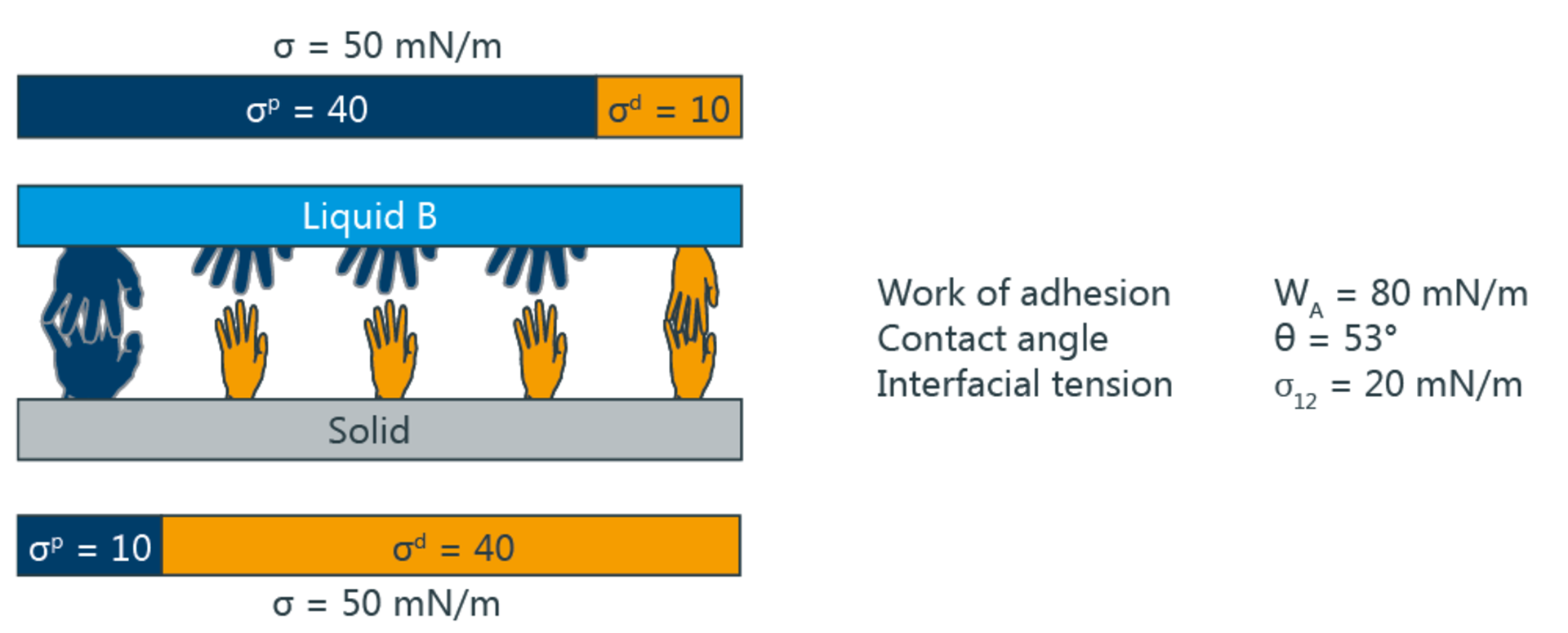
The pre-treatment methods mentioned above essentially increase the polar fraction of SFE and thus make the plastic more similar to water. According to the two-component model, wetting and adhesion are at a maximum if not only the SFE of the solid and the SFT of the liquid agree, but also the respective polar and disperse fractions.
The following illustration shows the wetting and adhesion depending on the polar and disperse fractions. The big hands symbolize polar, the small disperse interactions. In the upper picture, the interactions are identical, resulting in maximum adhesion and a contact angle of 0°. Below is an example with non-identical interaction components.
Which calculation models are available for the SFE?
Fowkes is the author of the first scientific paper in which the SFT of liquids was split into interaction fractions and the SFE was determined based on these fractions using contact angle measurements. The model most frequently used today is the one according to Owens, Wendt, Rabel and Kaelble (OWRK). It is based on Fowkes and uses contact angles of two liquids with known polar and disperse fractions of SFE. Other, less frequently used models interpret the polar and disperse fractions differently from OWRK or do not interpret the SFE and SFT at all in relation to their interaction fractions. The following table gives an overview; details on the models can be found in separate articles of this glossary.
| Model according to author(s) | Interaction components of the SFT |
|---|---|
| Fowkes | Disperse part and non-disperse part |
| Owens-Wendt-Rabel & Kaelble | Disperse and polar part |
| Wu | Disperse and polar part |
| Schultz | Disperse and polar part, measurement in bulk liquid phase |
| Oss, Good (acid-base) | Lewis acid part and Lewis base part |
| Extended Fowkes | Disperse and polar part and hydrogen bond part |
| Zisman | No division into components; determination of critical surface tension |
| Neumann Equation of State | No division into components |
Can't the SFE also be determined with test inks?
Ink tests are still used to check the wettability of surfaces. The result is interpreted as SFE by users and also by some test ink manufacturers. The method is seemingly obvious: several liquid mixtures with descending SFT are applied to the surface one after the other. The SFT of the first liquid, which forms a non-running film, i.e. completely wets the surface, is equated with the SFE of the solid. However, the nature of the interactions is not taken into account. As a consequence, the results of the ink test cannot be transferred to contact with most other liquids. In the technical process, the solid therefore often behaves quite differently than was to be expected after the ink test. The following example illustrates this: The polar water has a higher SFT than the non-polar diiodomethane (DIM), a standard liquid for SFE determination. On nonpolar plastics, water has a much higher contact angle than DIM. The ratio is reversed when the same liquids come into contact with clean glass as a highly polar surface: The water contact angle is considerably smaller than the DIM contact angle. This example shows that the amount of SFT of the liquid alone is not sufficient to establish a relationship between wetting and SFE. The published scientific study Why Test Inks Cannot Tell the Whole Truth about Surface Free Energy of Solids provides a comprehensive, critical discussion of the ink test method (see literature list).
Bibliography
- F. M. Fowkes, Attractive Forces at Interfaces. In: Industrial and Engineering Chemistry 56,12 (1964), P. 40-52.
- M. Jin, F. Thomsen, T. Skrivanek and T. Willers, Why Test Inks Cannot Tell the Whole Truth About Surface Free Energy of Solids. In: K. L. Mittal (Ed.), Advances in Contact Angle, Wettability and Adhesion Volume 2 , Hoboken, New Jersey and Salem, Massachusetts 2015, P. 419-438.
- D. H. Kaelble, Dispersion-Polar Surface Tension Properties of Organic Solids. In: J. Adhesion 2 (1970), P. 66-81.
- D. Owens; R. Wendt, Estimation of the Surface Free Energy of Polymers. In: J. Appl. Polym. Sci 13 (1969), P. 1741-1747.
- W. Rabel, Einige Aspekte der Benetzungstheorie und ihre Anwendung auf die Untersuchung und Veränderung der Oberflächeneigenschaften von Polymeren. In: Farbe und Lack 77,10 (1971), P. 997-1005.
- T. Young, An Essay on the Cohesion of Fluids. Philosophical Transactions of the Royal Society of London, The Royal Society, London 1805, Vol. 95, P. 65-87.






